Machine Translated by Google
Chemical solution coating on silicon substrate using Nano Dip Coater ND-0407-S3 and the effect of substrate lifting conditions
Sophia University, Faculty of Science and Technology, Department of Materials and Life Sciences
Hiroshi Uchida
1. Research background
The thin film fabrication method by coating a chemical solution made from organometallic compounds is complicated and requires large areas.This method is highly useful industrially because it allows for easy production of inorganic thin films on a substrate structure having the above properties.
In solution coating using a dip coater, not only the viscosity and hydrolysis behavior of the solution are important, In addition, the configuration of the experimental equipment has a large effect on the state of the coating thin film, and it is difficult to efficiently produce the desired thin film.
In order to obtain the desired results, it is necessary to precisely control these conditions.
Using a nano dip coater that can be precisely controlled, Thin film deposition experiments were verified.
2. Experimental Method
In this experiment, titanium tetra -n-butoxide monomer, which is frequently used in the preparation of titanium oxide (TiO2) thin films, was used.A 2-methoxyethanol solution (concentration 0.1 mol/dm3 ) was used as the chemical solution for coating.
The solution was dip-coated onto a Si wafer that had been mirror-polished on one side.
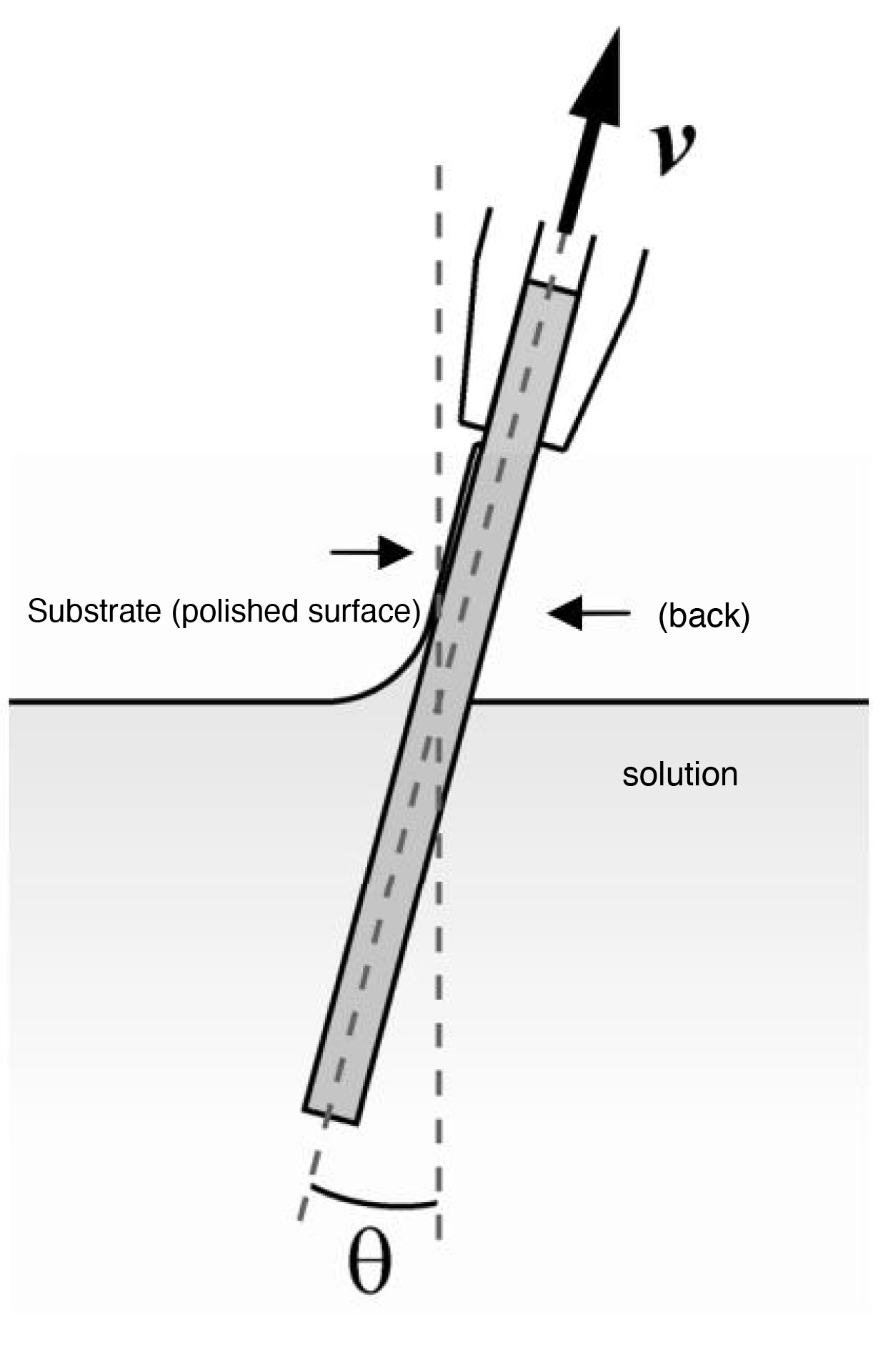
(ND-0407-S3, SDI Co., Ltd.) was used to pull the substrate at various pulling speeds (v) and angles (ÿ).
An outline of the substrate pulling operation and the pulling conditions are shown in Figure 1 and Table 1, respectively.
The deposition conditions were adjusted by heat-treating the precursor thin film coated on the plate (500°C, 5 min, in air).
Finally, TiO2 thin films with different crystallinity were obtained. The deposition conditions of the thin films were evaluated by photochemical analysis of the deposits on the polished surface.
The results were analyzed by optical and electron microscopy.
3. Results and Discussion
(1) Effect of substrate pulling speedThe deposition of thin films when the aforementioned chemical solutions were dip-coated on Si wafers at different withdrawal speeds was The stacking behavior was investigated (samples No. 1 to 3).
An optical microscope photograph of the deposit formed on the substrate surface is shown in Figure 2. The substrate was prepared at a pulling speed of 100.00 μm/s.
In the thin film sample (No. 1), almost no deposits were observed on the substrate surface.
The thin film samples (No. 2 and No. 3) prepared at 10.00 and 1.00 μm/s had glossy light brown and dark blue colors.
The formation of deposits was confirmed. In addition, the amount of fine particles, such as suspended dust, on the substrate was small.
It was increased by decreasing the deposition rate.
The cross-sectional structure of the deposit/substrate is shown in Fig. 3. The substrate was pulled up at a speed of 10.0 μm/s.
The thin film sample (No. 2) had a thickness of about 20 nm, and the thin film sample (No. 3) prepared at 1.00 μm/s had a plate-like deposit with a thickness of about 60 nm.
The presence of deposits was hardly observed in the case of (No. 1). These results are based on the results of dip coating.
This result is different from the previous report [1], which showed that the coating thickness increases with increasing substrate pulling speed.
This was due to the relatively low viscosity of the chemical solution used in this experiment and the slow withdrawal speed.
In the case of coating with a slow rate, the hydrolysis of the raw material at the substrate-solution interface proceeds sufficiently.
This is expected, but there is currently no confirmation.

 The color change is presumably a phenomenon that occurs due to changes in the state of interference of light waves between the surface reflected light of the thin film and the
interface reflected light (thin film/substrate) caused by an increase or decrease in the film thickness.
The color change is presumably a phenomenon that occurs due to changes in the state of interference of light waves between the surface reflected light of the thin film and the
interface reflected light (thin film/substrate) caused by an increase or decrease in the film thickness.(2) Effect of substrate lifting angle
Next, we investigated the thin film deposition behavior when the chemical solution was dip-coated on the Si wafer at different substrate pull- up angles (samples No. 3 to 6). The substrate pull-up speed was fixed at 100.00 μm/s, which was the angle at which the largest film thickness was obtained in the previous section.
Optical microscope photographs of the deposits formed on the substrate surface are shown in Figure 4. Blue deposits were observed in the thin film sample (No. 3) prepared at a pull-up angle of 0° (Figure 2), whereas light brown deposits were observed in the sample (No. 4) prepared at a pull-up angle of 5°. As the pull-up angle was increased, the color of the deposits became lighter, and at a pull-up angle of 30° (No. 6), the presence of deposits was barely noticeable. The amount of fine particles also decreased by increasing the substrate pull-up angle.
Electron microscope images of the deposit/substrate cross-sectional structures of these thin film samples are shown in Figure 5. A deposit with a thickness of approximately 15 nm was observed in the sample (No. 4) prepared with a substrate pull-up angle of 5o, and a deposit with a thickness of approximately 10 nm was observed in the thin film sample (No. 5) prepared with a substrate pull-up angle of 5o. Almost no deposit was observed in the thin film sample (No. 6) prepared with a substrate pull-up angle of 30o.

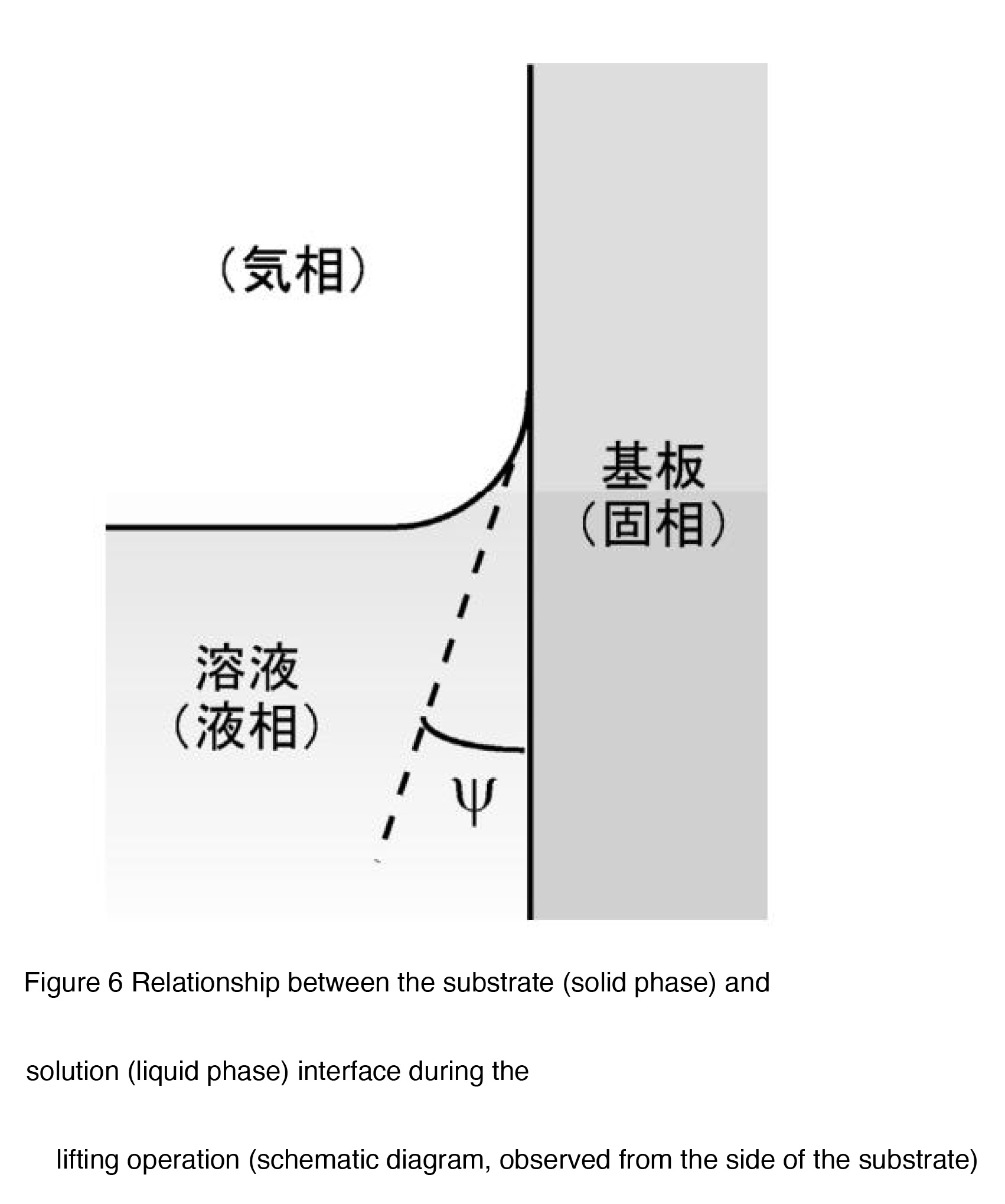
 By changing the substrate lifting angle, it is possible to change the contact state of the solution
on the substrate surface during dip coating, and in this experiment, we expect that the effect of
this is reflected in the thickness of the coating film. That is, a concave meniscus is generated at
the contact interface between the substrate (solid phase) and the solution (liquid phase), but by
increasing the substrate lifting angle from 0o , the contact angle (ÿ) between the solution and
substrate decreases [2], and as a result, the thickness of the liquid film formed on the substrate
surface during coating decreases.
By changing the substrate lifting angle, it is possible to change the contact state of the solution
on the substrate surface during dip coating, and in this experiment, we expect that the effect of
this is reflected in the thickness of the coating film. That is, a concave meniscus is generated at
the contact interface between the substrate (solid phase) and the solution (liquid phase), but by
increasing the substrate lifting angle from 0o , the contact angle (ÿ) between the solution and
substrate decreases [2], and as a result, the thickness of the liquid film formed on the substrate
surface during coating decreases.It is presumed that the above phenomenon occurs when the solution concentration is adjusted.
It is assumed that this also occurs when the liquid viscosity is changed.
4. Summary
Thin film samples were prepared by applying a chemical solution for TiO2 coating onto a Si wafer substrate under various substrate pulling conditions using a nano dip coater .As a result, it was confirmed in this experiment that the state of the deposit formed on the substrate changes significantly depending on the pulling speed and angle. Since the various parameters related to the substrate pulling conditions are factors closely related to the contact state between the substrate (solid phase) and the solution (liquid phase), it is expected that precise and diverse thin film deposition can be achieved by further controlling these parameters.* References:
[1] M.Guglielmi et al., J. Mater. Sci., 27, 5052 (1992). and many others [2] “Principles of Colloid and Surface Chemistry, 3rd Edition”, eds. P.C. Hiemenz and R. Rajagopalan (Marcel Dekker, New York, 1997) p.251.
 Japanease
Japanease English
English