Machine Translated by Google
March 30, 2007
Titanium oxide photocatalyst coating using Micro Dip Coater MD-0408
Naoya Yoshida, Jun Ogata, Toshiya Watanabe, International and University-
Industry Collaborative Research Center, The University of Tokyo
1. Introduction
Titanium oxide photocatalysts, discovered by Honda and Fujishima in the 1970s1 , have recently been attracting attention again as an environmental purification material, and many researches have been conducted on them, with many products now being commercially available2-4. They are broadly divided into two types of forms: powder and thin film. From the viewpoint of photocatalytic function, properties such as crystallinity are important, and for powders, the particle size, particle size distribution, and dispersibility as a colloid are important factors, while for thin films, the film thickness and uniformity are important factors. In fact, titanium oxide thin films are produced by various methods, but they can be broadly divided into two types: dry methods, such as sputtering processes, and wet methods using the sol- gel method5-6 . Although the dry method can be expected to achieve precise film thickness control and high uniformity, it is basically a vacuum process, making it an expensive system. On the other hand, the wet method is a simple process in which a precursor solution for the sol-gel method is applied to a substrate and then baked. In our laboratory, we conduct research using both processes, and the wet method has many advantages, such as the comparative ease of changing the composition of the thin film, the ease of constructing a laminated film, and the ability to apply it to substrates with complex shapes, making the dip coating method a useful film formation method along with spin coating and spray coating. Titanium alkoxides (e.g., titanium(IV) tetraisopropoxide, etc.) are often used as titanium oxide precursors for coating, and coating solutions with improved stability and coating properties are commercially available from several manufacturers. Here, a study was carried out on titanium oxide photocatalyst coating by the wet method using a micro dip coater MD-0408 with the aim of uniform film formation and thickness control.
2.coating solution.
A mirror-polished silicon wafer was used as the substrate. After solvent cleaning and UV-ozone treatment, the substrate was quickly dip-coated using 10 ... Experiment Nippon Soda Bistrater NDH-510C was used as the During dip coating, the dependence of film thickness and uniformity on the pulling speed was examined, and the repetitive coating properties were examined with a fixed pulling speed of 4 mm/sec. Film thickness was measured using a spectroscopic ellipsometer (JA Woollam, The surface roughness was calculated by fitting to a model of Si/SiO2/ TiO2(/surface roughness) using a VASE system . The surface shape was measured using a laser microscope (Keyence Corporation, The measurements were performed using a microscope (VF-7510) and an AFM (Keyence, VN-8010).3. Results and Discussion
Problems that often arise in the film formation process using the sol- gel method are the stability and viscosity of the precursor solution and the volatility of the solvent. To form a uniform film, it is often necessary to operate quickly in a dry atmosphere such as synthetic air. For this reason, we installed a micro dip coater in a simple glove box. In addition, we modified the position of the dip coater motor and also made a custom-made hanger. Since it is difficult to reach the top of the dip coater and remove the substrate inside the glove box, we use a custom-made product as shown in Figure 1 to make it easy and quick to operate. As a result, smooth and high-quality film formation is possible. quickly dip-coated using 10 ...
Experiment Nippon Soda Bistrater NDH-510C was used as the
Figure 1. Custom-made hanger
In the case of film formation by the dip coating method, the thickness of the film can
be controlled by changing either the concentration and viscosity of the coating solution
or the pulling speed. However, there are many restrictions on the coating solution and
it is often difficult to change it significantly, so the pulling speed is often the only option.
It is realistic to deal with this problem by controlling the pulling speed.
Furthermore, in reality, there is a limit to how much the pulling speed can
be controlled, and as the pulling speed is increased in order to deposit a
thicker film, it becomes difficult to obtain a uniform film, and so it is
necessary to combine it with repeated coating, as described below.
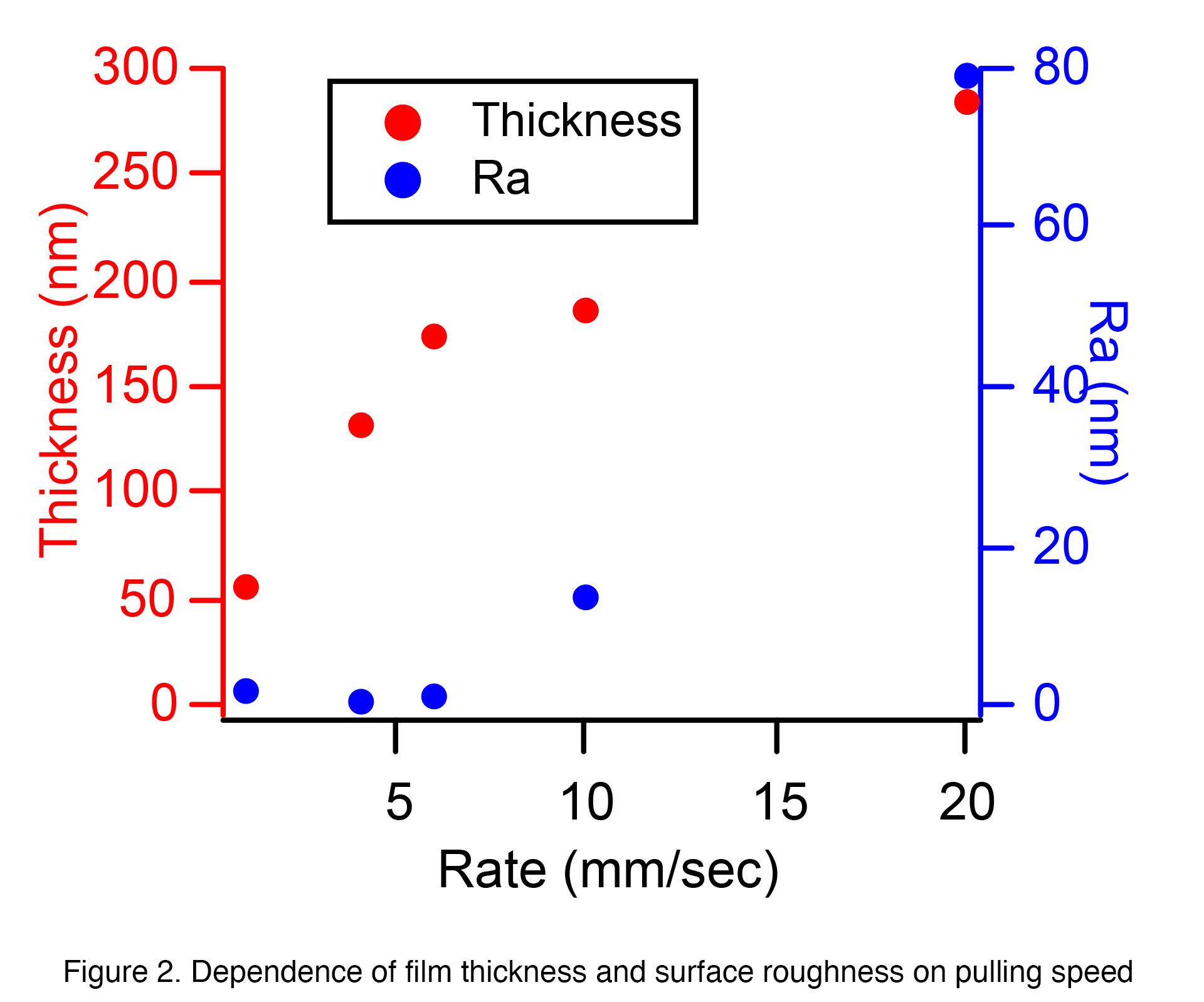
Therefore, we first investigated the dependency on the pulling speed. As
shown in Figure 2, at a pulling speed of about 6 mm/sec or less, it was
possible to control the film thickness (approximately 180 nm or less)
without changing the surface roughness (Ra). However, at a pulling speed of 10 mm/sec or more,
The thickness and thickness of the film did not increase monotonically, and the increase
in surface roughness was noticeable. In particular, when the speed was increased to 20
mm/sec, there was a noticeable uneven coating (due to shrinkage of the liquid film during
the drying process immediately after dip coating) and numerous cracks appeared after baking.
quickly dip-coated using 10 ...
Experiment Nippon Soda Bistrater NDH-510C was used as the
Figure 1. Custom-made hanger
In the case of film formation by the dip coating method, the thickness of the film can
be controlled by changing either the concentration and viscosity of the coating solution
or the pulling speed. However, there are many restrictions on the coating solution and
it is often difficult to change it significantly, so the pulling speed is often the only option.
It is realistic to deal with this problem by controlling the pulling speed.
Furthermore, in reality, there is a limit to how much the pulling speed can
be controlled, and as the pulling speed is increased in order to deposit a
thicker film, it becomes difficult to obtain a uniform film, and so it is
necessary to combine it with repeated coating, as described below.
Therefore, we first investigated the dependency on the pulling speed. As
shown in Figure 2, at a pulling speed of about 6 mm/sec or less, it was
possible to control the film thickness (approximately 180 nm or less)
without changing the surface roughness (Ra). However, at a pulling speed of 10 mm/sec or more,
The thickness and thickness of the film did not increase monotonically, and the increase
in surface roughness was noticeable. In particular, when the speed was increased to 20
mm/sec, there was a noticeable uneven coating (due to shrinkage of the liquid film during
the drying process immediately after dip coating) and numerous cracks appeared after baking.
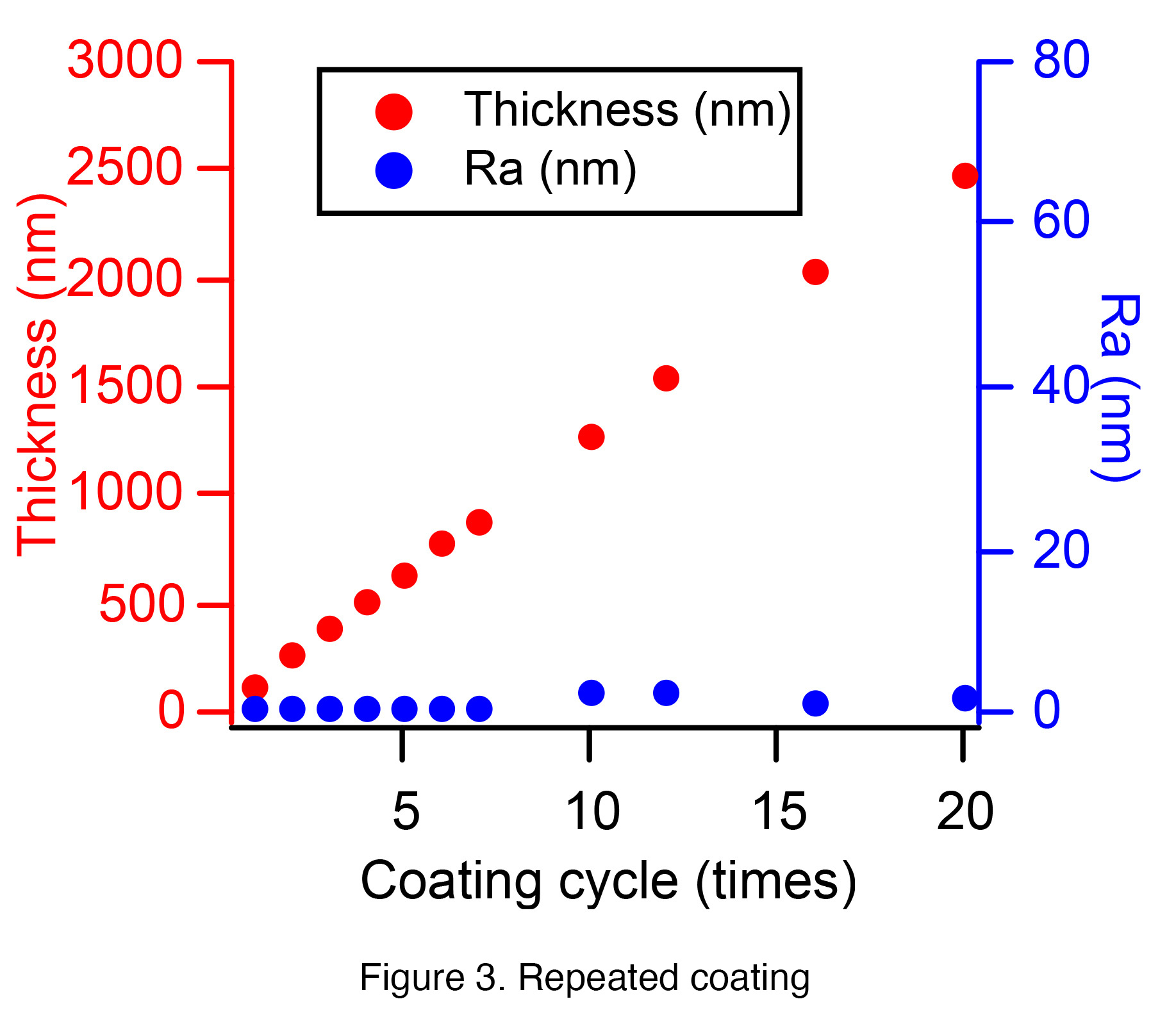
 Next, repeated coating was investigated. The pulling speed was fixed at 4 mm/sec, and
dip coating and baking were counted as one cycle. Film formation was performed up to
20 cycles, and the film thickness and surface roughness were measured. The film
thickness at a pulling speed of 4 mm/sec was about 130 nm, and a nice linear relationship
was obtained with respect to the number of repetitions, as shown in Figure 3. On the
other hand, the surface roughness (Ra) hardly changed, indicating that a good film was
formed.
Next, repeated coating was investigated. The pulling speed was fixed at 4 mm/sec, and
dip coating and baking were counted as one cycle. Film formation was performed up to
20 cycles, and the film thickness and surface roughness were measured. The film
thickness at a pulling speed of 4 mm/sec was about 130 nm, and a nice linear relationship
was obtained with respect to the number of repetitions, as shown in Figure 3. On the
other hand, the surface roughness (Ra) hardly changed, indicating that a good film was
formed.
 Japanease
Japanease English
English